
Each time the water freezes, the crack gets bigger, but both pieces are still sandstone. An example might be ice expanding a crack on the side of an arch. Physical weathering breaks the rock into smaller pieces, but the components remain virtually the same. There are two types of weathering: physical and chemical. This confusion is understandable because they are often intertwined. Weathering is the physical breakup of rocks, and is often confused with erosion, which is the removal of rock by gravity, wind, and water once it has weathered. UV light is also monitored, which indirectly reflects the condition of the upper atmosphere ozone layer. 
Even if all countries quit using CFCs, however, they will linger in the upper atmosphere for decades. 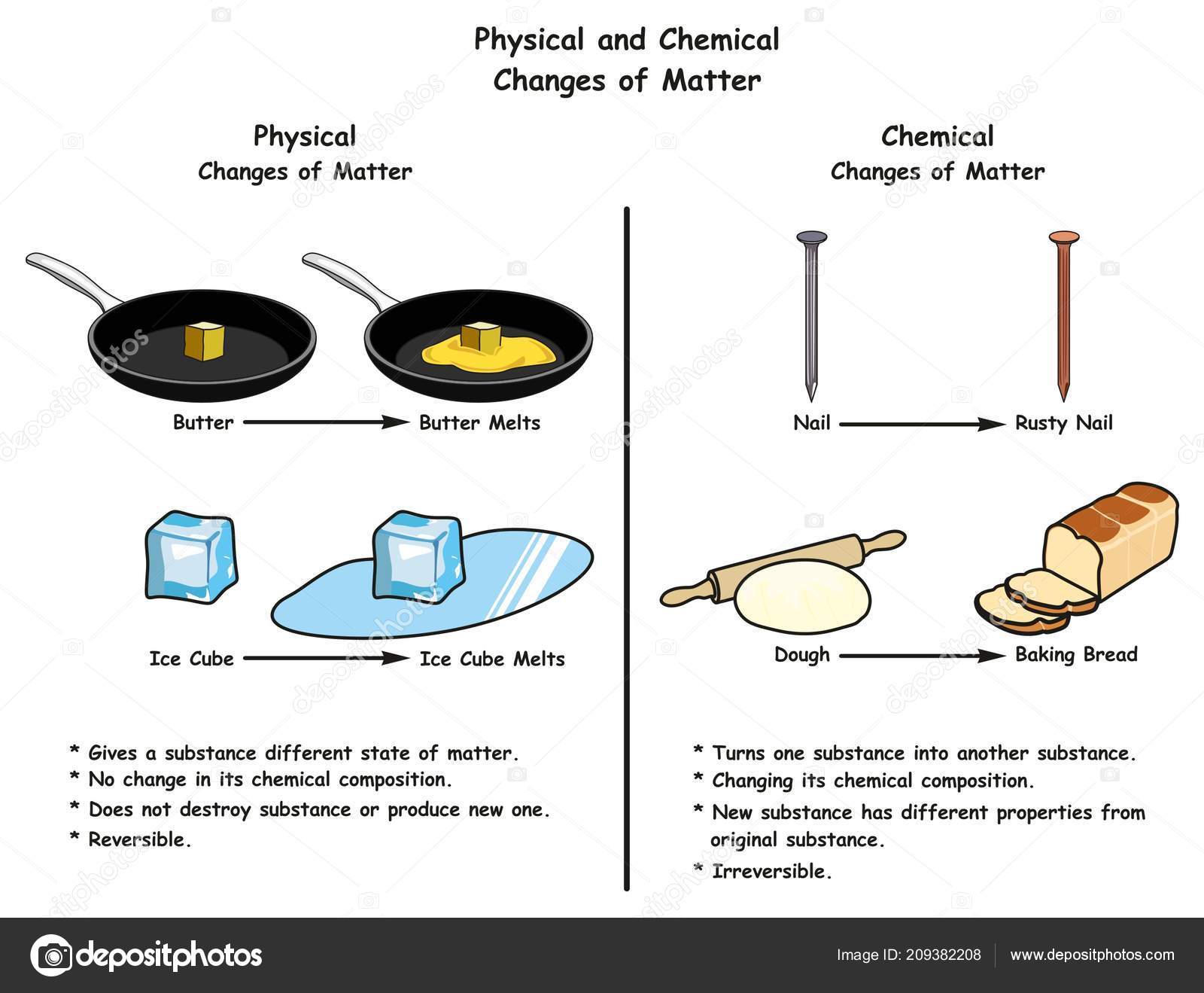
Since 1987, The Montreal Protocol outlawed the use of CFCs in the United States and many countries. Humans used CFCs to manufacture hundreds of different products, including Styrofoam packaging, aerosol spray cans, and as the coolants in refrigerators and air conditioners. This reaction is a chemical change in the ozone molecule. The single largest factor in the destruction of the ozone layer is a family of chemicals called chlorofluorocarbons (CFCs). But, increased UV light from a damaged ozone layer leads to increased incidences of skin and eye disease in humans as well as damage to some wildlife and plants. Normal quantities of UV light are good for such things as plant growth and suntans. The stratospheric ozone layer blocks much of the sun’s UV light from reaching the earth’s surface. 
Unlike in the lower atmosphere, Ozone plays a positive role in the upper atmosphere. Scientists monitor ozone levels to provide real time data on air quality to the public. Canyonlands National Park also monitors ozone at ground level. Car exhaust, the result of a chemical change in fuel, is a major contributor of ozone to the lower atmosphere. High levels of ozone in the lower atmosphere can cause human health problems and can contribute to the greenhouse effect. Ozone is an invisible gas made of 3 oxygen atoms. Burning a piece of paper would be a chemical change, as would baking a cake. At the end of a chemical change, you have a new substance. Chemical changes are those where one or more substances are combined to produce a new substance. For example, chopping up a carrot or ice melting into water are both physical changes. Physical changes are those in which the shape, size, or state of the matter changes, but the substance is still essentially the same. Matter can exist as a solid, liquid, or gas (or plasma) and can change in many ways. Air, water, rocks, trees, stars, and animals all consist of matter. Everything that has mass and volume, no matter how small, is made of matter. Emphasize whether a new substance is or is not created by the formation of a new substance with different properties. Standard 5.2.3 Plan and carry out investigations to determine the effect of combining two or more substances. Emphasize making observations of changes supported by a particle model of matter. Standard 5.2.1 Develop and use a model to describe that matter is made of particles on a scale that is too small to be seen. Whether a change results in a new substance or not, the total amount of matter is always conserved. When two or more different substances are combined a new substance with different properties may be formed. Substances have specific properties by which they can be identified. Matter is made of particles that are too small to be seen but still exist and can be detected by other means. Strand 5.2: PROPERTIES AND CHANGES OF MATTER All substances are composed of matter. Utah Science with Engineering Education Standards: Strand 5.2: Properties and Changes of Matter:Įssential Question: What physical and chemical changes in matter can be observed in nature? Temperature changes (either the temperature increases or decreases).Grade Level: Upper Elementary: Third Grade through Fifth Grade Subject: Science State Standards: Utah Science Standards.Observations that help to indicate chemical change include: Some of the evidence for chemical change will involve the energy changes that occur in chemical changes, but some evidence involves the fact that new substances with different properties are formed in a chemical change. We have to make other observations to indicate that a chemical change has happened. We can't actually see molecules breaking and forming bonds, although that's what defines chemical changes. \): Burning of wax to generate water and carbon dioxide is a chemical reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
